
Director of Research
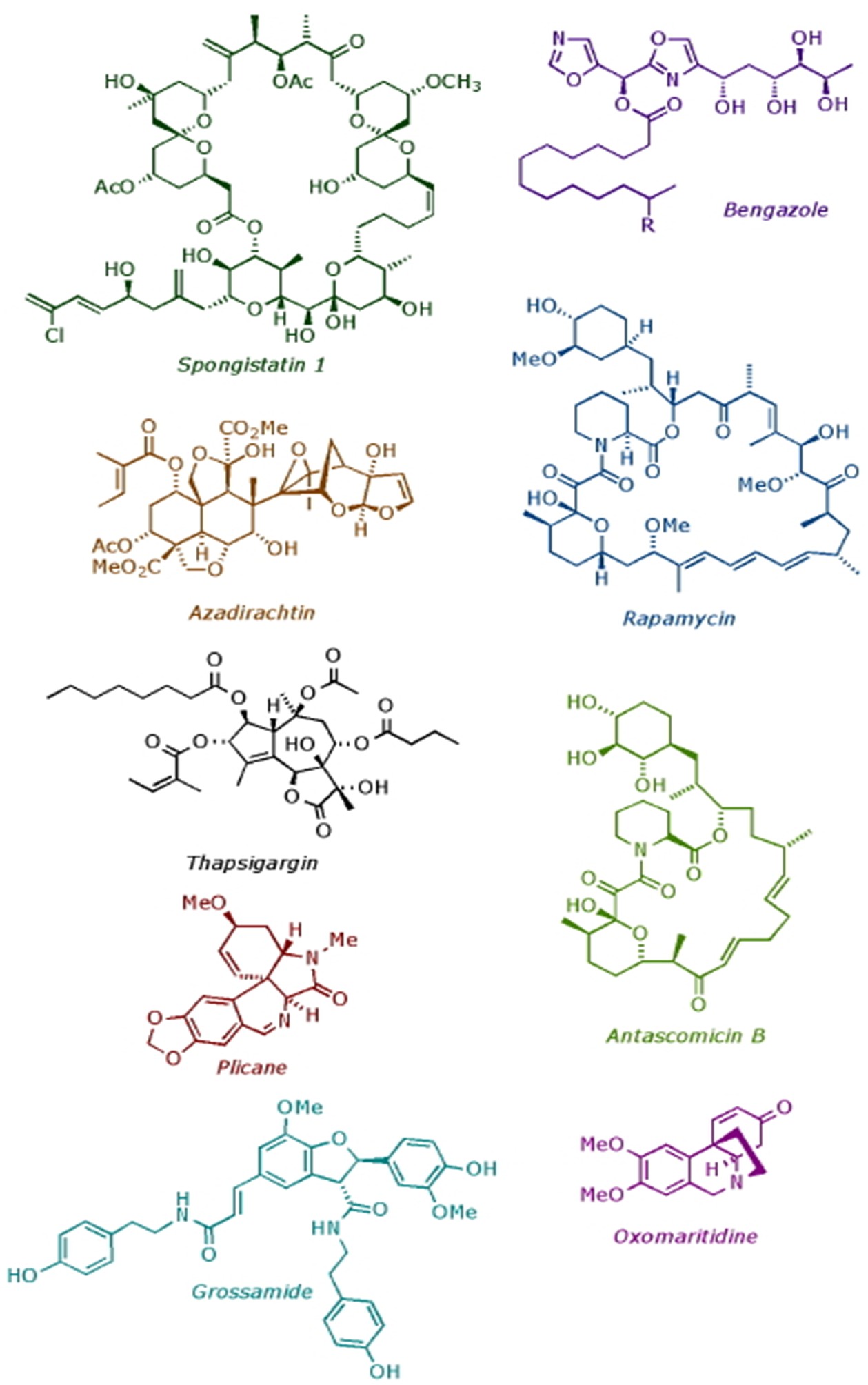
In the Ley Group, we specialise in developing new synthesis methods and applying them to the construction of biologically important molecules. Over the years we have completed the total synthesis of many natural products, including: spongistatin 1 (anti-mitotic agent); rapamycin (immunosuppressant); thapsigargin (SERCA pumps inhibitor); azadirachtin (insect antifeedant); and bengazole A (fungicide). In addition to our research on natural product synthesis, we also pioneered flow chemistry and machine assisted synthesis.
For more detailed research information and our publication list, please see our legacy group website.
Completed Natural Products
Publications
The Evolution of Flow Chemistry: An Opinion on Factors Driving Innovation.
Chimia (Aarau)
(2023)
77
288
(doi: 10.2533/chimia.2023.288)
Exploring the chemical space of phenyl sulfide oxidation by automated optimization
Reaction Chemistry and Engineering
(2023)
8
538
(doi: 10.17863/CAM.93992)
Multicomponent Direct Assembly of N-Heterospirocycles Facilitated by Visible-Light-Driven Photocatalysis.
J Org Chem
(2022)
87
13204
(doi: 10.1021/acs.joc.2c01684)
Automated multi-objective reaction optimisation: which algorithm should I use?
Reaction Chemistry & Engineering
(2022)
7
987
(doi: 10.1039/d1re00549a)
Photoredox-Catalyzed Dehydrogenative Csp3–Csp2 Cross-Coupling of Alkylarenes to Aldehydes in Flow
The Journal of Organic Chemistry
(2021)
86
13559
(doi: 10.1021/acs.joc.1c01621)
Formation and utility of reactive ketene intermediates under continuous flow conditions
Tetrahedron
(2021)
93
132305
(doi: 10.1016/j.tet.2021.132305)
CLICK-enabled analogues reveal pregnenolone interactomes in cancer and immune cells
Iscience
(2021)
24
102485
(doi: 10.1016/j.isci.2021.102485)
- ‹ previous
- Page 2

