
Teaching Professor of Chemistry
I am a Teaching Professor in Inorganic Chemistry in the Department of Chemistry and the Deputy Senior Tutor at Churchill College. My teaching duties include lecturing courses on the structure, bonding and reactivity of transition metal complexes and on electron deficient, main group compounds. I demonstrate in the practical classes and conduct small group tutorials (supervisions) for chemistry students from all four years of the Natural Sciences degree. I encourage students to think laterally about the subject and to make connections between topics through collaborative discussion and linking theoretical chemistry work to practical sessions.
Research interests
My research focusses on the synthesis of heavier transition metal organometallic species for conjugation to protein targets via specific, controlled placement. The aim is to construct and evolve artificial metalloenzymes in order to explore catalytic activity and deliver activity, selectivity and new-to-nature reactivity.
Organometallic cofactors
Many natural metalloenzymes assemble from proteins and biosynthesised complexes, generating potent catalysts.
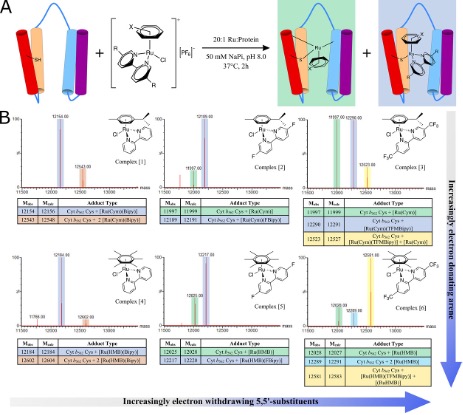
Work in our group to generate artificial metalloenzymes (ArMs) uses ligand exchange to unmask catalytic activity. Heavier transition metal complexes are designed with sacrificial ligands to generate new enzymes via ligand exchange in proteins. The resulting metal cofactors form direct, peptidic coordination bonds between metal complexes and protein scaffolds but also retain non-biological ligand(s). We use tandem mass spectrometry and 19F NMR spectroscopy to characterise the organometallic cofactors and identify the protein-derived ligands.
We have generated catalytically active ArMs and are working towards artificially evolving these, like their natural counterparts, for new-to-nature reactivity and improved catalytic activity and selectivity.
Controlled Ligand Exchange Between Ruthenium Organometallic Cofactor Precursors and a Naïve Protein Scaffold Generates Artificial Metalloenzymes Catalysing Transfer Hydrogenation
Publications
- Page 1

