Professor of Materials Chemistry
Email: acf50 [at] cam.ac.uk
Phone: 01223 336368
Open positions: We are interested to hear from prospective PhD and MPhil applicants, please email Alex if you are interested. We are committed to supporting diversity, equity and inclusion. Applications to the group are particularly encouraged from BAME students, women, students with diverse gender identities, and other underrepresented groups. We are also committed to supporting safe and sustainable working practices.
Overview: Our ultimate goal in the Forse group is to understand and improve materials that can reduce greenhouse gas emissions and tackle the global warming crisis. Limiting global warming to 1.5 ºC requires the rapid development and deployment of a range of greenhouse gas mitigation technologies. In the Forse group we explore the functional behaviour of new materials for CO2 capture and electrochemical energy storage. We specialise in the application of nuclear magnetic resonance (NMR) spectroscopy techniques that are complemented by synthetic chemistry, electrochemistry and computational chemistry.
Click here for a full list of publications.
Electrochemical CO2 Capture. An alternative approach to traditional CO2 capture by absorbents, which use thermal energy to release CO2, is to use electrical energy to perform the capture-release cycle. This has the potential to achieve a more energy-efficient capture process, though information is lacking on the molecular level mechanisms of capture, making optimisation difficult. We are combining in situ experimental methods (electrochemistry, gas adsorption, spectroscopy) with computational chemistry to unravel the mechanisms of electrochemical CO2 capture and to explore the use of electricity for other chemical separations.
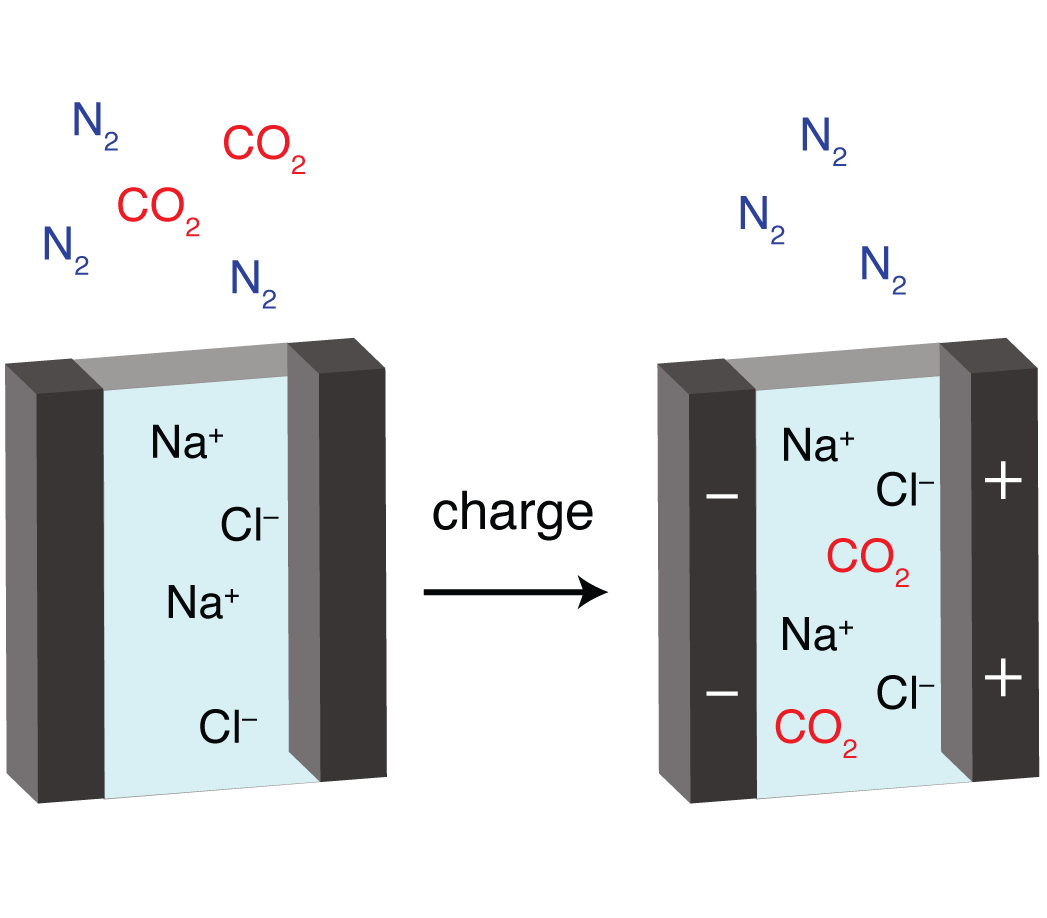
Improved Energy Storage. We are exploring supercapacitors and batteries for the efficient storage of renewable energy. We study the fundamentals of ionic adsorption and diffusion processes in electrodes, electrolytes and separators so that improved materials can be designed and synthesised. We combine chemical synthesis and electrochemical characterization with mechanistic studies by spectroscopy and molecular simulation. Through these studies we establish design principles for improved energy storage devices.
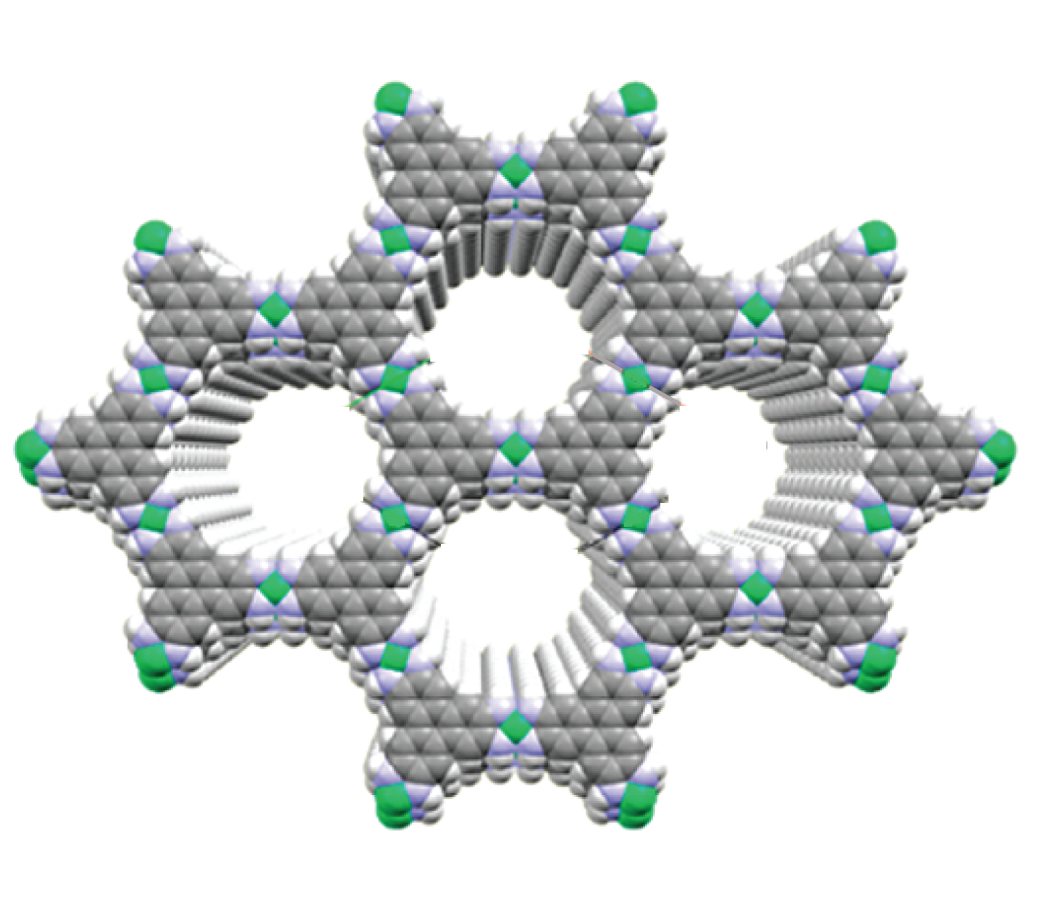
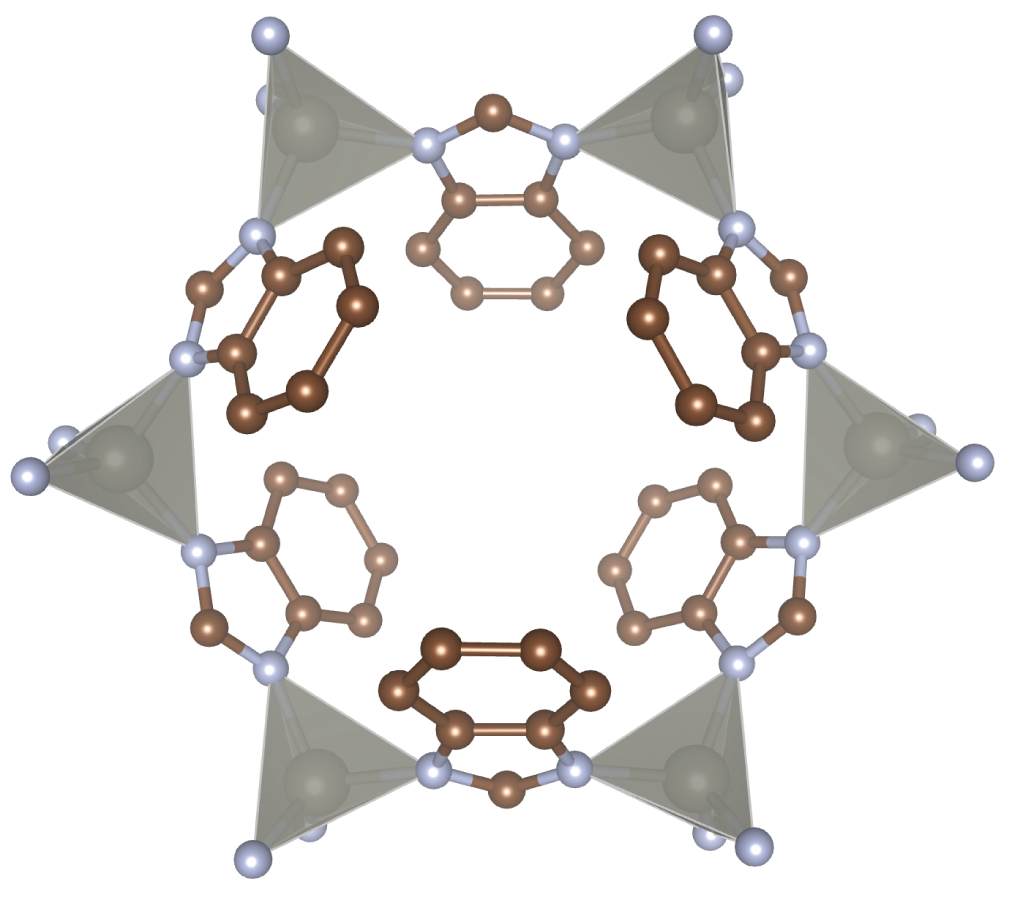
New Materials for Separating Molecules. Membranes and adsorbent materials have the potential to perform CO2 capture and other molecular separations in a more energy-efficient way. However, the design of improved materials is hampered by a lack of atomic-scale characterization techniques that probe the conditions relevant to the real separation process. We are tackling this issue by developing in situ spectroscopic methods to study the functioning behaviour of materials for molecular separations. We combine chemical synthesis, spectroscopy and molecular simulations to devise materials with improved performance.