
Director of Research
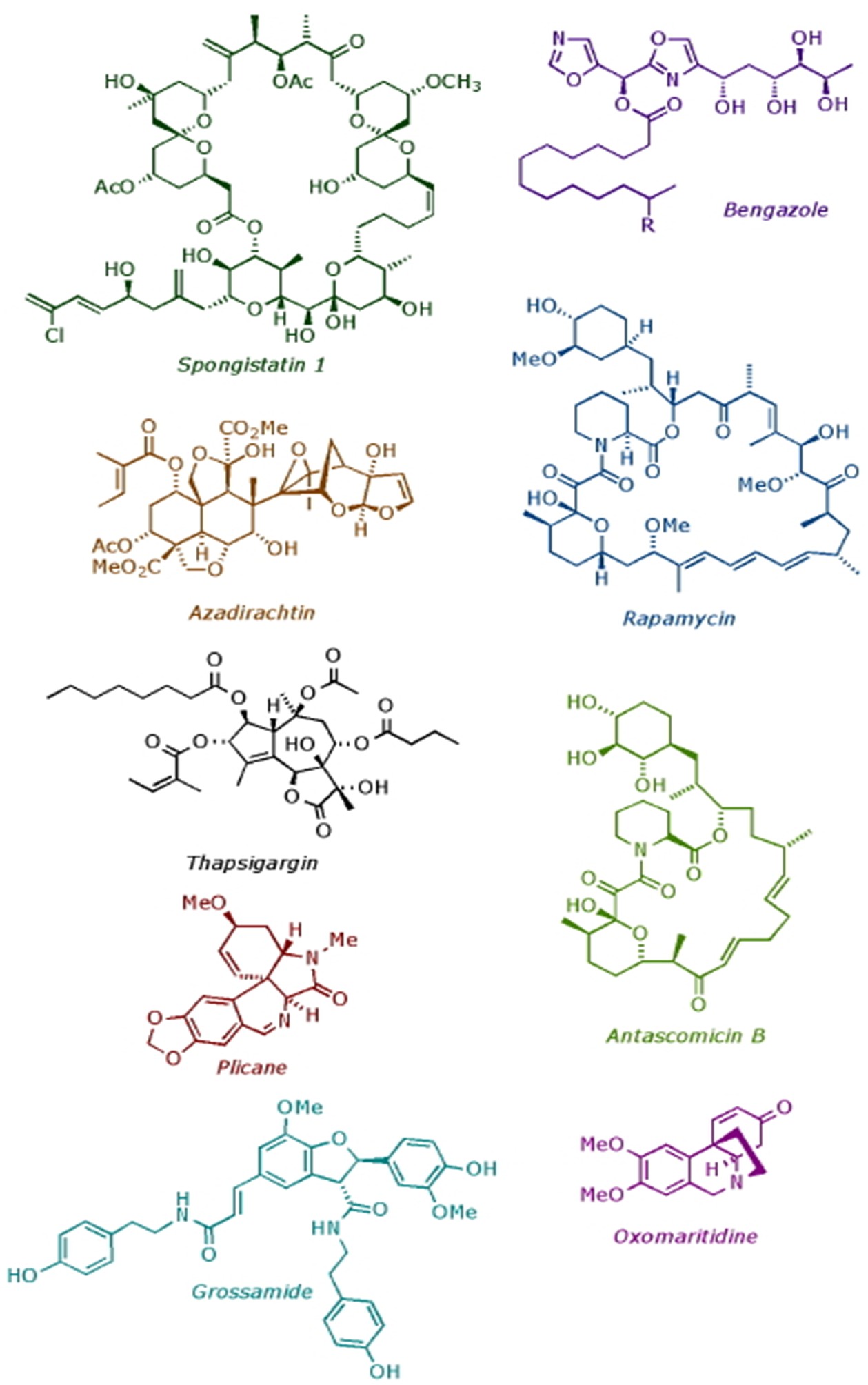
In the Ley Group, we specialise in developing new synthesis methods and applying them to the construction of biologically important molecules. Over the years we have completed the total synthesis of many natural products, including: spongistatin 1 (anti-mitotic agent); rapamycin (immunosuppressant); thapsigargin (SERCA pumps inhibitor); azadirachtin (insect antifeedant); and bengazole A (fungicide). In addition to our research on natural product synthesis, we also pioneered flow chemistry and machine assisted synthesis.
For more detailed research information and our publication list, please see our legacy group website.
Completed Natural Products
Publications
ENABLING METHODOLOGY - THE SYNTHETIC CHEMISTS CONTRIBUTION TO MOLECULAR RECOGNITION
MOLECULAR RECOGNITION : CHEMICAL AND BIOCHEMICAL PROBLEMS II
(1992)
111
183
Dispiroketals in synthesis: Preparation of a stable, sterically demanding glyceraldehyde ketal and diastereoselective reactions with simple organometallic reagents
Synthesis
(1992)
1992
52
(doi: 10.1055/s-1992-34153)
FURTHER REACTIONS OF TERT-BUTYL 3-OXOBUTANTHIOATE AND TERT-BUTYL 4-DIETHYL-PHOSPHONO-3-OXOBUTANTHIOATE - CARBONYL COUPLING REACTIONS, AMINATION, USE IN THE PREPARATION OF 3-ACYLTETRAMIC ACIDS AND APPLICATION TO THE TOTAL SYNTHESIS OF FULIGORUBIN-A
Tetrahedron
(1992)
48
1145
Chemistry of insect antifeedants from Azadirachta indica (part 12): use of silicon as a control element in the synthesis of a highly functionalized decalin fragment of azadirachtin
J CHEM SOC PERK T 1
(1992)
2735
(doi: 10.1039/P19920002735)
The use of π-allyltricarbonyliron lactone complexes in the synthesis of the β-lactone esterase inhibitor (−)-valilactone.
Tetrahedron
(1991)
47
9929
Synthesis of the alkaloids (−)-heliotridane and (−)-isoretronecanol via π-allyltricarbonyliron lactam complexes.
Tetrahedron Letters
(1991)
32
7119
(doi: 10.1016/0040-4039(91)85056-b)
Chemistry of insect antifeedants from Azadirachta indica (part 11): Characterisation and structure activity relationships of some novel rearranged azadirachtins.
Tetrahedron
(1991)
47
9231
CHEMISTRY OF INSECT ANTIFEEDANTS FROM AZADIRACHTA-INDICA .10. SYNTHESIS OF A HIGHLY FUNCTIONALIZED DECALIN FRAGMENT OF AZADIRACHTIN
Tetrahedron Letters
(1991)
32
6187
(doi: 10.1016/0040-4039(91)80785-5)
CHEMISTRY OF INSECT ANTIFEEDANTS FROM AZADIRACHTA-INDICA .9. OXIDATIVE REACTIONS OF AZADIRACHTIN DERIVATIVES LEADING TO C8-C14 BOND-CLEAVAGE
Tetrahedron Letters
(1991)
32
6183
(doi: 10.1016/0040-4039(91)80784-4)
- ‹ previous
- Page 83

