
Director of Research
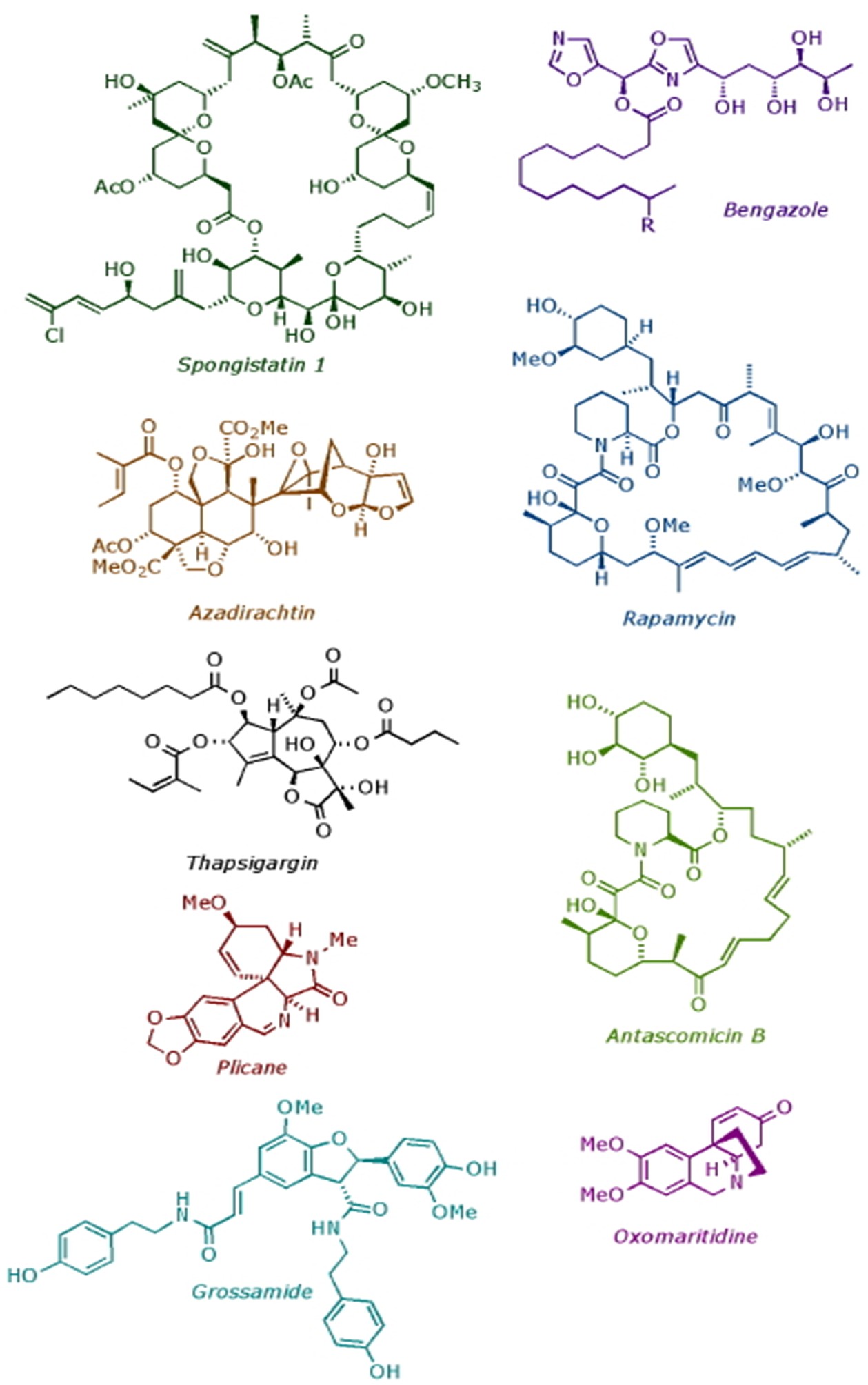
In the Ley Group, we specialise in developing new synthesis methods and applying them to the construction of biologically important molecules. Over the years we have completed the total synthesis of many natural products, including: spongistatin 1 (anti-mitotic agent); rapamycin (immunosuppressant); thapsigargin (SERCA pumps inhibitor); azadirachtin (insect antifeedant); and bengazole A (fungicide). In addition to our research on natural product synthesis, we also pioneered flow chemistry and machine assisted synthesis.
For more detailed research information and our publication list, please see our legacy group website.
Completed Natural Products
Publications
STEREOCHEMISTRY OF A 5,5,10-TRIMETHYL-2-DECALONE BY PROTON NUCLEAR MAGNETIC-RESONANCE - A NEW APPLICATION OF INDOR DIFFERENCE SPECTROSCOPY
J CHEM RES-S
(1983)
210
The total synthesis of the clerodane diterpene insect antifeedant ajugarin I
Chemical Communications
(1983)
503
(doi: 10.1039/c39830000503)
Peterson olefination using phenylsulphonyltrimethylsilylmethane. A new preparation of vinylic sulphones
Journal of the Chemical Society Chemical Communications
(1983)
1281
(doi: 10.1039/c39830001281)
Total synthesis of the structurally unique ionophore antibiotic X-14547 A
J. Chem. Soc., Chem. Commun.
(1983)
630
(doi: 10.1039/c39830000630)
Synthesis of methyl-1,6-dioxaspiro[4,5]decanes using organoselenium mediated cyclization reactions
Tetrahedron Letters
(1982)
23
4625
A conformational study of elaiophylin by X-ray crystallography and difference 1H NMR methods; observation of a selective sign reversal of the nuclear overhauser effect
Tetrahedron Letters
(1982)
23
1207
The X-ray structure and absolute configuration of insect antifeedant clerodane diterpenoids from Teucrium africanum
JOURNAL OF THE CHEMICAL SOCIETY-PERKIN TRANSACTIONS 1
(1982)
1005
(doi: 10.1039/p19820001005)
Thermal rearrangement reactions of tricarbonyliron lactone complexes
Journal of the Chemical Society Perkin Transactions 1
(1982)
1355
(doi: 10.1039/p19820001355)
Dehydrogenation of lactones using benzeneseleninic anhydride. X-ray crystal structure of 3β-acetoxy-14α-hydroxy-17a-oxa-D-homo-5α-androst- 15-en-17-one
Journal of the Chemical Society Perkin Transactions 1
(1982)
1919
(doi: 10.1039/p19820001919)
A practical catalytic method for the preparation of steroidal 1,4-dien-3-ones by oxygen atom transfer from iodoxybenzene to diphenyl diselenide
J CHEM SOC PERK T 1
(1982)
1947
(doi: 10.1039/p19820001947)
- ‹ previous
- Page 96

