
Director of Research
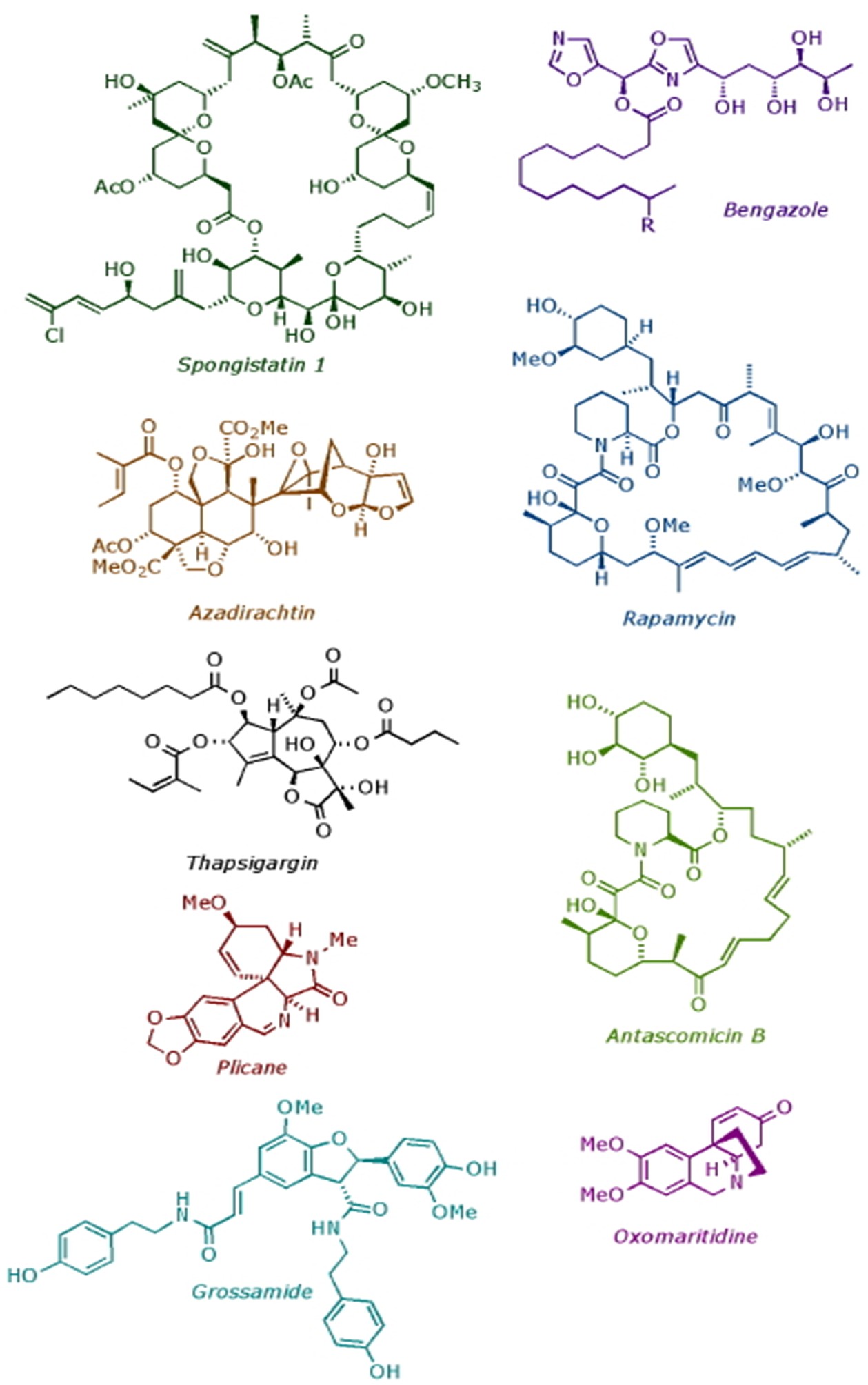
In the Ley Group, we specialise in developing new synthesis methods and applying them to the construction of biologically important molecules. Over the years we have completed the total synthesis of many natural products, including: spongistatin 1 (anti-mitotic agent); rapamycin (immunosuppressant); thapsigargin (SERCA pumps inhibitor); azadirachtin (insect antifeedant); and bengazole A (fungicide). In addition to our research on natural product synthesis, we also pioneered flow chemistry and machine assisted synthesis.
For more detailed research information and our publication list, please see our legacy group website.
Completed Natural Products
Publications
Dispiroketals in synthesis (part 10): Further reactions of dispoke protected lactate and glycolate enolates
Tetrahedron
(1994)
50
7157
Dispiroketals in synthesis (Part 5): A new opportunity for oligosaccharide synthesis using differentially activated glycosyl donors and acceptors
Tetrahedron Letters
(1993)
34
8523
Selective Acylation and Alkylation Reactions of Diols Using Dibutyltin Dimethoxide
Synlett
(1993)
1993
913
(doi: 10.1055/s-1993-22650)
TOTAL SYNTHESIS OF THE SPIROKETAL MACROLIDE (+) MILBEMYCIN ALPHA(1)
Tetrahedron Letters
(1993)
34
7479
Synthesis of 4-Substituted Imidazoles via 4-Metallo Imidazole Intermediates
Synlett
(1993)
1993
748
(doi: 10.1055/s-1993-22594)
Microwave Promoted Hydrolysis of Esters Absorbed on Alumina: A New Deprotection Method for Pivaloyl Groups
Synlett
(1993)
1993
793
(doi: 10.1055/s-1993-22612)
DISPIROKETALS IN SYNTHESIS .4. ENANTIOSELECTIVE DESYMMETRIZATION OF GLYCEROL USING A C2-SYMMETRICAL DISUBSTITUTED BIS-DIHYDROPYRAN
Tetrahedron Letters
(1993)
34
5649
Chemistry of insect antifeedants from Azadirachta Indica (Part 15):1 Degradation studies of Azadirachtin leading to C8C14 bond cleavage.
Tetrahedron
(1993)
49
1675
Dispiroketals in Synthesis (Part 3):1Selective Protection of Diequatorial Vicinal Diols in Carbohydrates
Synthesis
(1993)
1993
689
(doi: 10.1055/s-1993-25923)
- ‹ previous
- Page 81

