
Director of Research
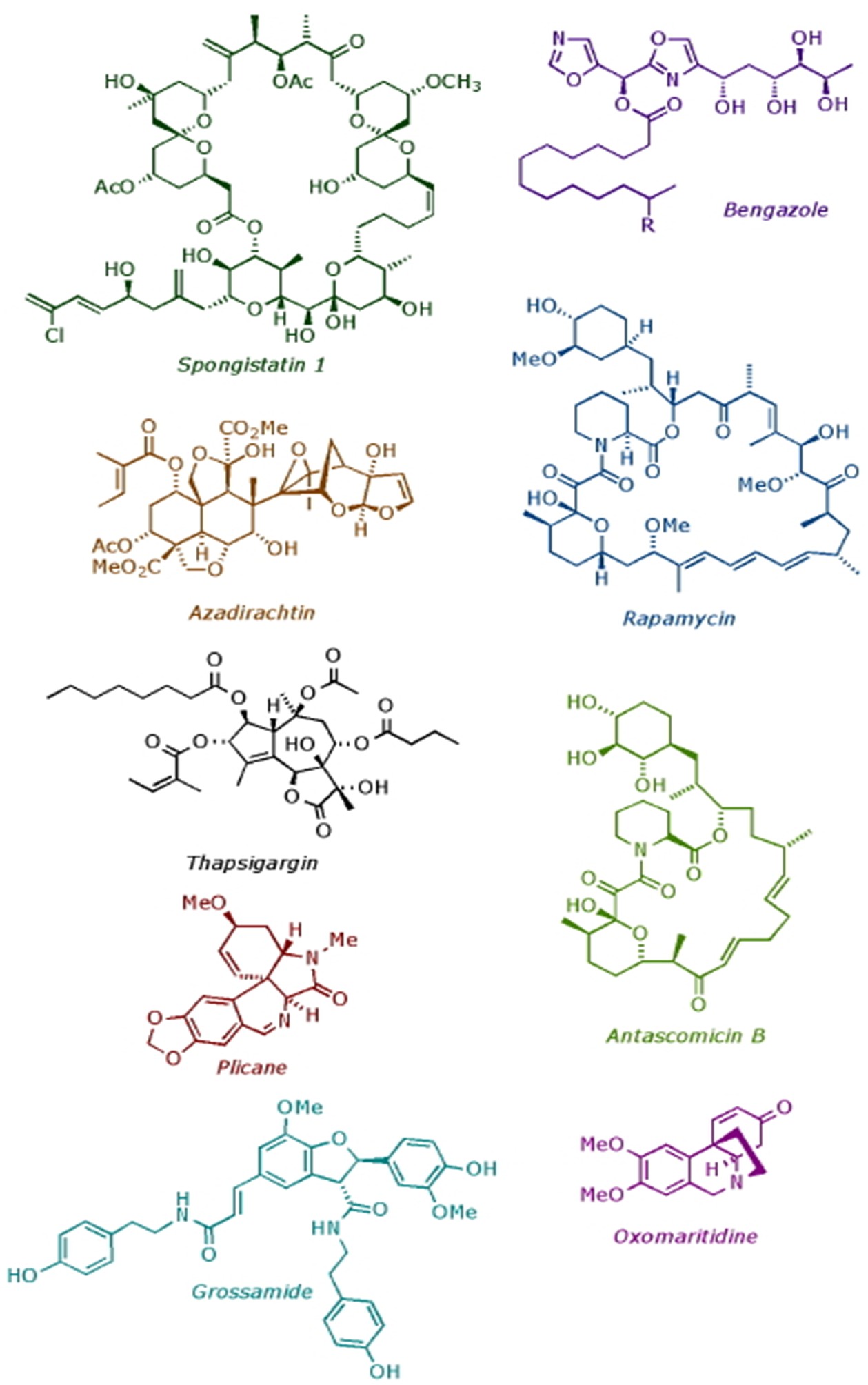
In the Ley Group, we specialise in developing new synthesis methods and applying them to the construction of biologically important molecules. Over the years we have completed the total synthesis of many natural products, including: spongistatin 1 (anti-mitotic agent); rapamycin (immunosuppressant); thapsigargin (SERCA pumps inhibitor); azadirachtin (insect antifeedant); and bengazole A (fungicide). In addition to our research on natural product synthesis, we also pioneered flow chemistry and machine assisted synthesis.
For more detailed research information and our publication list, please see our legacy group website.
Completed Natural Products
Publications
The total synthesis of the annonaceous acetogenin, muricatetrocin C
Chemistry
(2002)
8
1621
Analytical construct resins for analysis of solid-phase chemistry
Chemistry - A European Journal
(2002)
8
1768
The simultaneous use of immobilised reagents for the one-pot conversion of alcohols to carboxylic acids
JOURNAL OF THE CHEMICAL SOCIETY-PERKIN TRANSACTIONS 1
(2002)
2
1024
(doi: 10.1039/b201776h)
The use of pi-allyltricarbonyliron lactone complexes in the synthesis of the resorcylic macrolides alpha- and beta-zearalenol
J CHEM SOC PERK T 1
(2002)
2
874
(doi: 10.1039/b201164f)
Total Synthesis of the Cyclic Heptapeptide Argyrin B: A New Potent Inhibitor of T-Cell Independent Antibody Formation
Org Lett
(2002)
4
711
(doi: 10.1021/ol017184m)
The effects of phytochemical pesticides on the growth of cultured invertebrate and vertebrate cells
Pest Manag Sci
(2002)
58
268
(doi: 10.1002/ps.449)
Application of polymer-supported enzymes and reagents in the synthesis of γ-aminobutyric acid (GABA) analogues
Synlett
(2002)
1641
(doi: 10.1055/s-2002-34249)
A mild, enantioselective synthesis of (R)-salmeterol via sodium borohydride-calcium chloride asymmetric reduction of a phenacyl phenylglycinol derivative
J CHEM SOC PERK T 1
(2002)
2
2237
(doi: 10.1039/b207068p)
Reductive decomplexation of π-allyltricarbonyliron lactone complexes; a new route to stereo-defined 1,7-diols and 2,3-diene-1,7-diols
Chemical Communications
(2002)
2
2130
(doi: 10.1039/b206406p)
- ‹ previous
- Page 59

