
Director of Research
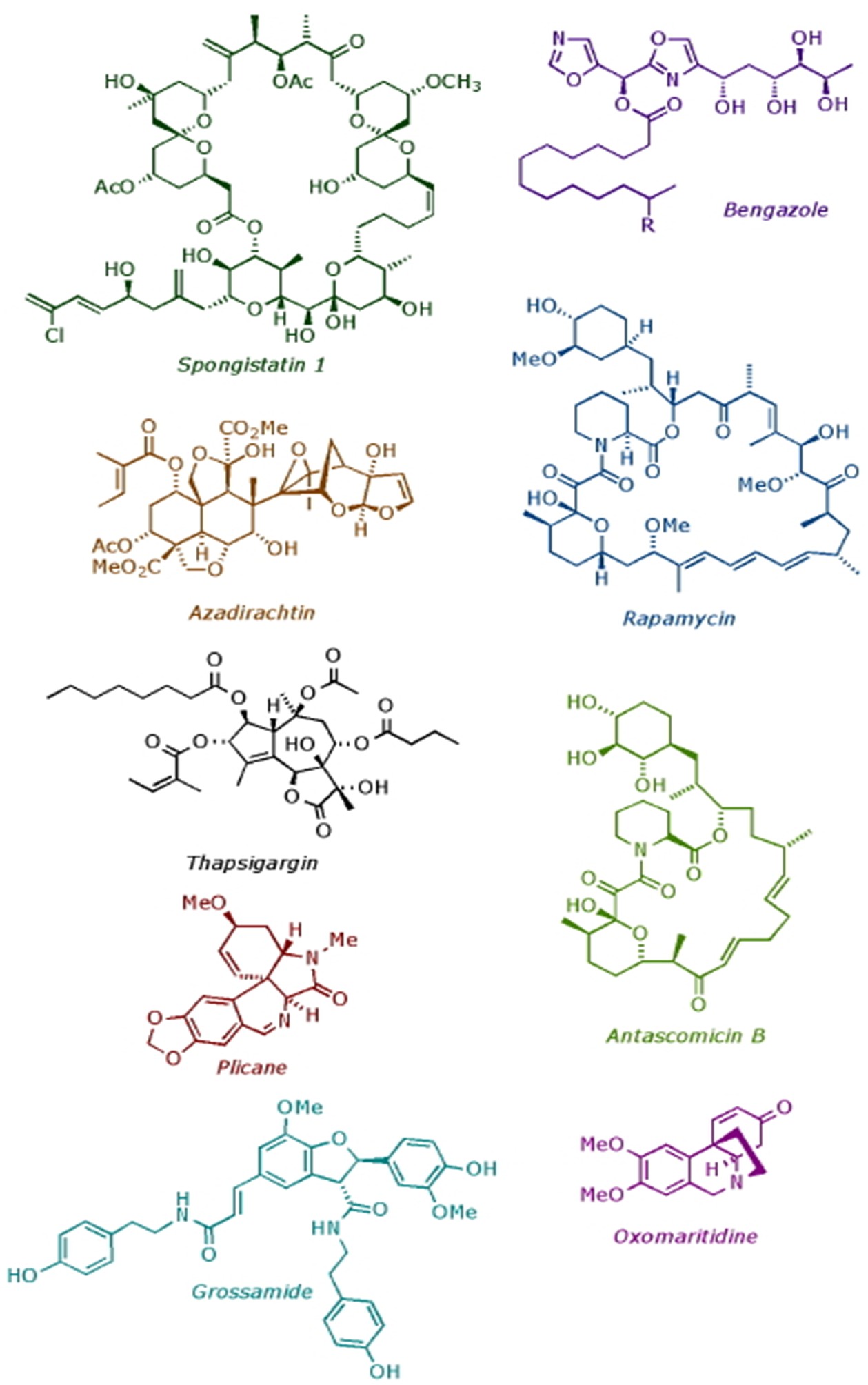
In the Ley Group, we specialise in developing new synthesis methods and applying them to the construction of biologically important molecules. Over the years we have completed the total synthesis of many natural products, including: spongistatin 1 (anti-mitotic agent); rapamycin (immunosuppressant); thapsigargin (SERCA pumps inhibitor); azadirachtin (insect antifeedant); and bengazole A (fungicide). In addition to our research on natural product synthesis, we also pioneered flow chemistry and machine assisted synthesis.
For more detailed research information and our publication list, please see our legacy group website.
Completed Natural Products
Publications
Data mining and the future of chemistry
MANUFACTURING CHEMIST
(2001)
72
14
1,2-diacetals: a new opportunity for organic synthesis.
Chemical Reviews
(2000)
101
53
(doi: 10.1021/cr990101j)
A rapid approach for the optimisation of polymer supported reagents in synthesis
Synlett
(2000)
2000
1603
(doi: 10.1055/s-2000-7917)
The synthesis of mono- and bicyclic ethers via acid catalysed ring-opening cyclisation of tetrahyclropyranyl ether derivatives
J CHEM SOC PERK T 1
(2000)
1829
(doi: 10.1039/a909302h)
Multi-step organic synthesis using solid-supported reagents and scavengers: A new paradigm in chemical library generation
Journal of the Chemical Society Perkin Transactions 1
(2000)
3815
(doi: 10.1039/b006588i)
Diastereoselective oxygen to carbon rearrangements of anomerically linked enol ethers and the total synthesis of (+)-(S,S )-(cis-6-methyltetrahydropyran-2-yl)acetic acid, a component of civet
J CHEM SOC PERK T 1
(2000)
2385
(doi: 10.1039/b001243m)
The use of pi-allyltricarbonyliron lactone complexes in the synthesis of the resorcylic macrolides alpha- and beta-zearalenol
J CHEM SOC PERK T 1
(2000)
3028
(doi: 10.1039/b005942k)
Methyl 2,3-O-(6,6′-octahydro-6,6′-bi-2h-pyran-2,2′-diyl)-α-d- galactopyranoside: α-D-Galactopyranoside, methyl, 2,3-O-octahydro[2,2′-bi-2H-pyran]-2,2′-diyl-, [2(2R,2′R)-]
Organic Syntheses
(2000)
77
212
1,7-Asymmetric induction of chirality in a Mukaiyama aldol reaction using π-allyltricarbonyliron lactone complexes: Highly diastereoselective synthesis of π-substituted β-hydroxy carbonyl compounds
J CHEM SOC PERK T 1
(2000)
1677
(doi: 10.1039/b002056g)
Oxygen to carbon rearrangements of anomerically linked alkenols from tetrahydropyran derivatives: An investigation of the reaction mechanism via a double isotopic labelling crossover study
Journal of the Chemical Society Perkin Transactions 1
(2000)
1815
(doi: 10.1039/a909300a)
- ‹ previous
- Page 63

