
Director of Research
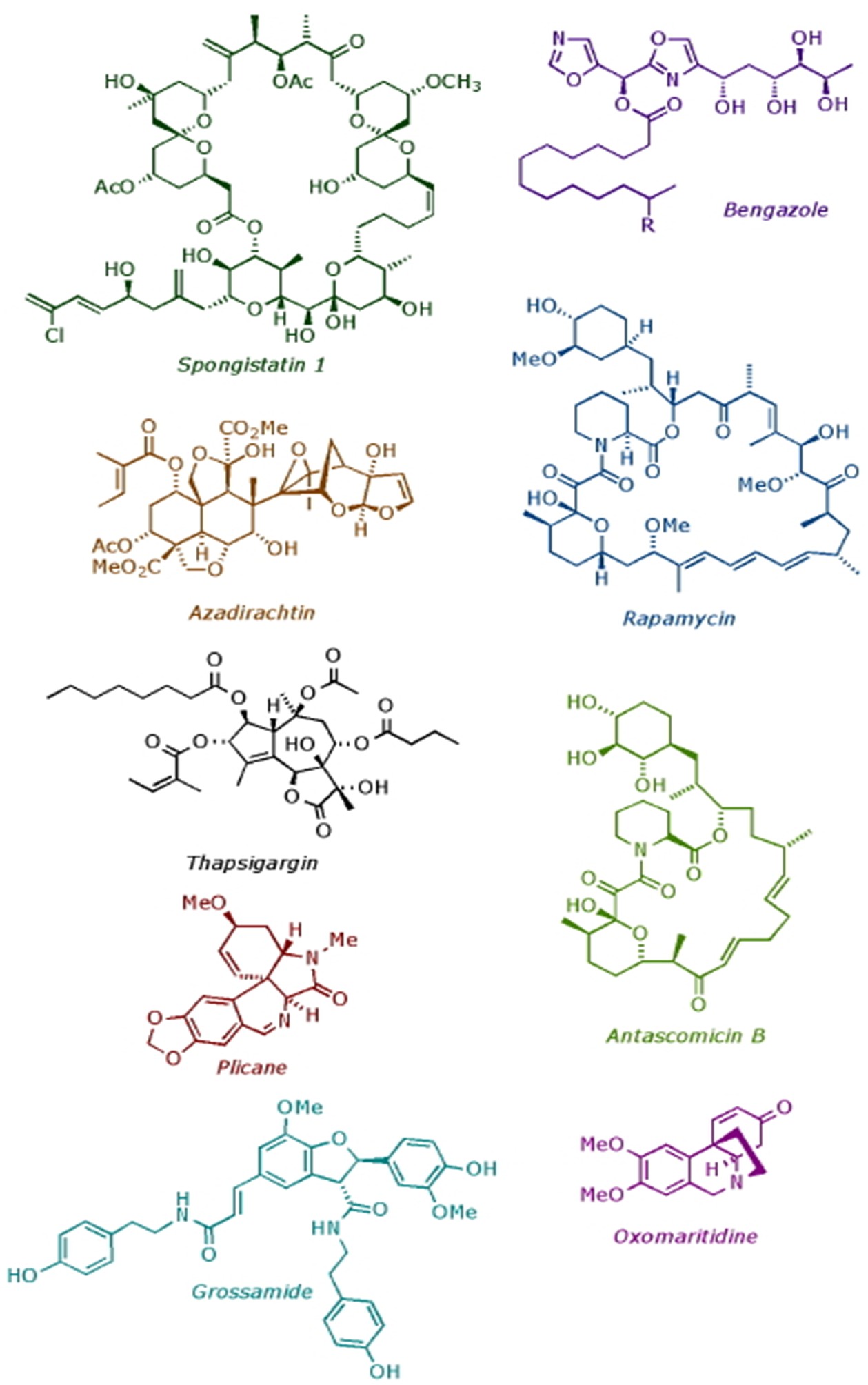
In the Ley Group, we specialise in developing new synthesis methods and applying them to the construction of biologically important molecules. Over the years we have completed the total synthesis of many natural products, including: spongistatin 1 (anti-mitotic agent); rapamycin (immunosuppressant); thapsigargin (SERCA pumps inhibitor); azadirachtin (insect antifeedant); and bengazole A (fungicide). In addition to our research on natural product synthesis, we also pioneered flow chemistry and machine assisted synthesis.
For more detailed research information and our publication list, please see our legacy group website.
Completed Natural Products
Publications
Synthesis of spongistatin 2 employing a new route to the EF fragment
Chemical Science
(2013)
4
1989
(doi: 10.1039/c3sc50304f)
The integration of flow reactors into synthetic organic chemistry
Journal of Chemical Technology & Biotechnology
(2013)
88
519
(doi: 10.1002/jctb.4012)
Synthesis of (-)-hennoxazole A: Integrating batch and flow chemistry methods
Synlett
(2013)
24
514
(doi: 10.1055/s-0032-1318109)
A Continuous Flow Solution to Achieving Efficient Aerobic Anti-Markovnikov Wacker Oxidation
Advanced Synthesis and Catalysis
(2013)
342
Flow chemistry synthesis of zolpidem, alpidem and other GABA A agonists and their biological evaluation through the use of in-line frontal affinity chromatography
Chemical Science
(2013)
4
764
(doi: 10.1039/c2sc21850j)
A prototype device for evaporation in batch and flow chemical processes
Green Chemistry
(2013)
15
2050
(doi: 10.1039/c3gc40967h)
Flow Chemistry Syntheses of Styrenes, Unsymmetrical Stilbenes and Branched Aldehydes
ChemCatChem
(2012)
5
159
(doi: 10.1002/cctc.201200778)
The synthesis of Bcr-Abl inhibiting anticancer pharmaceutical agents imatinib, nilotinib and dasatinib
Org Biomol Chem
(2012)
11
1766
(doi: 10.1039/c2ob27003j)
An expeditious synthesis of imatinib and analogues utilising flow chemistry methods
Organic & biomolecular chemistry
(2012)
11
1822
(doi: 10.1039/c2ob27002a)
Synthesis and Use of a Trifluoromethylated Azomethine Ylide Precursor
J Org Chem
(2012)
77
11071
(doi: 10.1021/jo302052m)
- ‹ previous
- Page 20

