
Director of Research
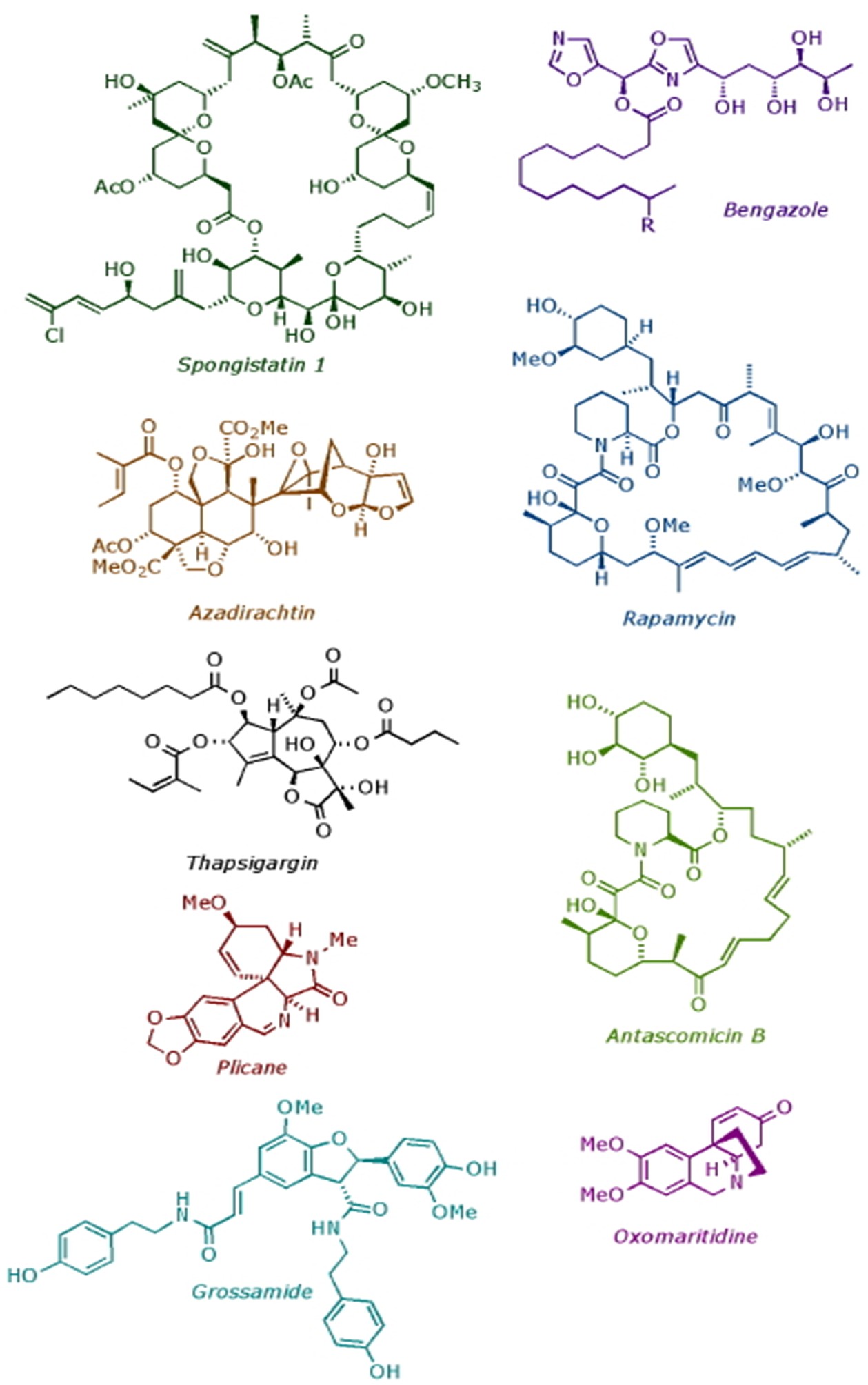
In the Ley Group, we specialise in developing new synthesis methods and applying them to the construction of biologically important molecules. Over the years we have completed the total synthesis of many natural products, including: spongistatin 1 (anti-mitotic agent); rapamycin (immunosuppressant); thapsigargin (SERCA pumps inhibitor); azadirachtin (insect antifeedant); and bengazole A (fungicide). In addition to our research on natural product synthesis, we also pioneered flow chemistry and machine assisted synthesis.
For more detailed research information and our publication list, please see our legacy group website.
Completed Natural Products
Publications
Some Alkylation and Reductive Amination Studies of b-Ketomacrolides as Potential Metal Speciation Materials
Heterocycles
(1993)
35
263
(doi: 10.3987/com-92-s12)
UPTAKE, RETENTION, METABOLISM AND EXCRETION OF [22,23-H-3(2)]DIHYDROAZADIRACHTIN IN SCHISTOCERCA-GREGARIA
Journal of Insect Physiology
(1993)
39
935
(doi: 10.1016/0022-1910(93)90003-A)
Microbial Oxidation in Synthesis: Preparation of a Potential Insulin Mimic from Benzene
Synlett
(1992)
1992
997
(doi: 10.1055/s-1992-21561)
Alfred bader [2]
Chemical and Engineering News
(1992)
70
2
Chemistry of insect antifeedants from Azadirachta indica(part 12): use of silicon as a control element in the synthesis of a highly functionalized decalin fragment of azadirachtin
Journal of the Chemical Society Perkin Transactions 1
(1992)
2735
(doi: 10.1039/p19920002735)
Chemistry of insect antifeedants from Azadirachta indica, Part 14: Absolute configuration of azadirachtin
Chemical Communications
(1992)
1304
(doi: 10.1039/C39920001304)
Total synthesis of the ionophore antibiotic CP-61,405 (routiennocin)
Tetrahedron
(1992)
48
7899
Dispiroketals in synthesis (part 2): A new group for the selective protection of diequatorial vicinal diols in carbohydrates.
Tetrahedron Letters
(1992)
33
4767
BADER,ALFRED
CHEMICAL & ENGINEERING NEWS
(1992)
70
2
Total Synthesis of the Carboxylic Acid lonophore Antibiotic CP-61,405 (Routiennocin): Preparation of the Inherent Spiroketal Unit via a Reverse Coupling Process
Synlett
(1992)
1992
399
(doi: 10.1055/s-1992-21358)
- ‹ previous
- Page 82

