
Director of Research
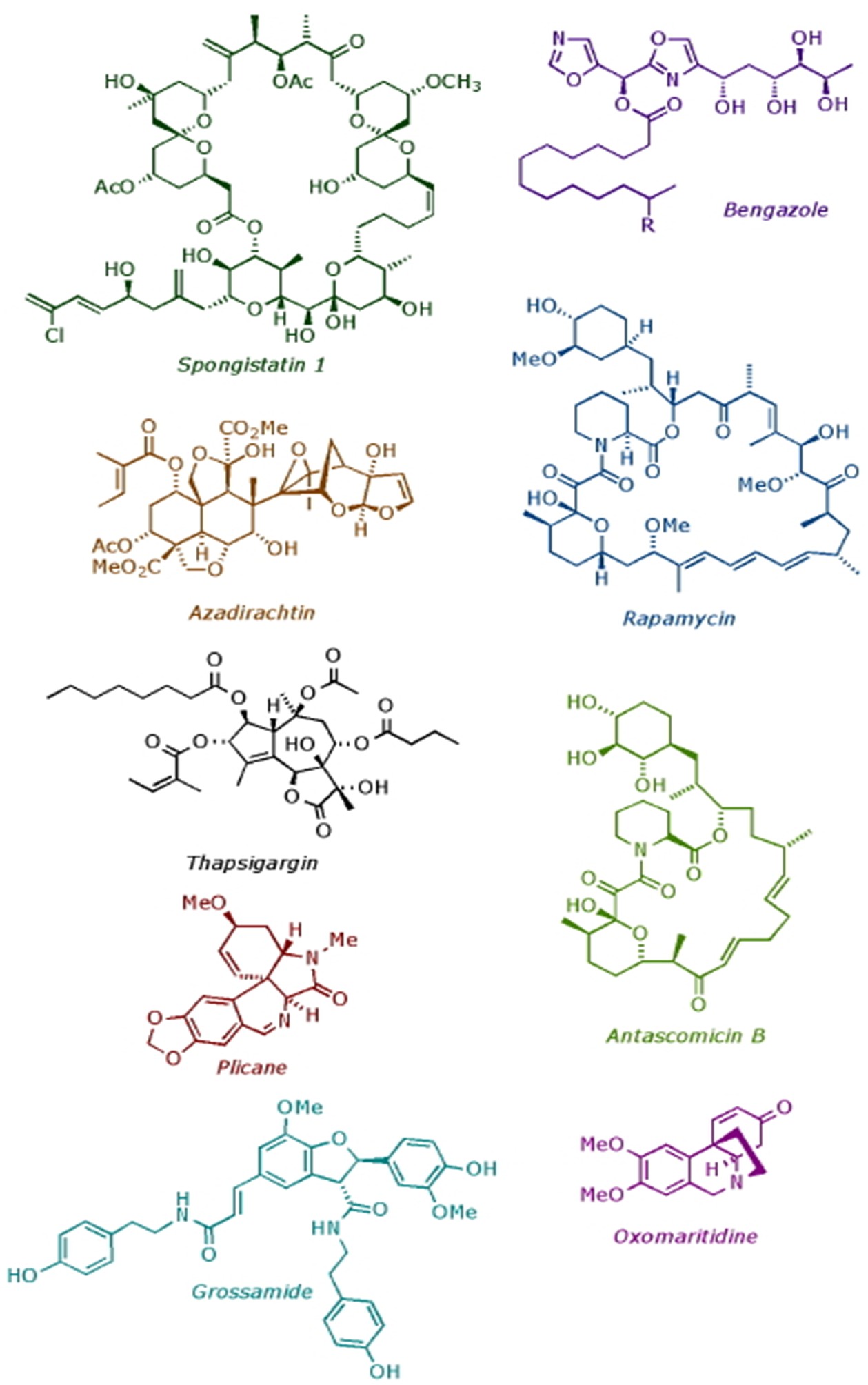
In the Ley Group, we specialise in developing new synthesis methods and applying them to the construction of biologically important molecules. Over the years we have completed the total synthesis of many natural products, including: spongistatin 1 (anti-mitotic agent); rapamycin (immunosuppressant); thapsigargin (SERCA pumps inhibitor); azadirachtin (insect antifeedant); and bengazole A (fungicide). In addition to our research on natural product synthesis, we also pioneered flow chemistry and machine assisted synthesis.
For more detailed research information and our publication list, please see our legacy group website.
Completed Natural Products
Publications
Development of β-keto 1,3-dithianes:: Versatile platforms for organic synthesis.
ABSTRACTS OF PAPERS OF THE AMERICAN CHEMICAL SOCIETY
(2003)
225
U342
Toward a total synthesis of thapsigargin.
ABSTRACTS OF PAPERS OF THE AMERICAN CHEMICAL SOCIETY
(2003)
225
U351
Organic‐Catalyst‐Mediated Cyclopropanation Reaction
Angewandte Chemie (International ed. in English)
(2003)
42
828
(doi: 10.1002/anie.200390222)
Transfer hydrogenation using recyclable polyurea-encapsulated palladium: Efficient and chemoselective reduction of aryl ketones
Chem Commun (Camb)
(2003)
3
678
(doi: 10.1039/b300074p)
Reductive decomplexation of pi-allyltricarbonyliron lactone complexes using sodium naphthalenide as a route to stereodefined 1,7-diols and 2,3-diene-1,7-diols
Org Biomol Chem
(2003)
1
3197
(doi: 10.1039/b306861g)
Synthesis of Enantiomers ofButane-1,2-diacetal-Protected Glyceraldehyde and of (R,R)-Butane-1,2-diacetal-ProtectedGlycolic Acid
Synthesis
(2003)
1598
(doi: 10.1055/s-2003-40519)
Total Synthesis of the PhytotoxicAgent Herbarumin II Using Butane Diacetals of Glycolic Acid as BuildingBlocks
Synlett
(2003)
1186
(doi: 10.1055/s-2003-39892)
Copper(I)-catalyzed preparation of (E)-3-iodoprop-2-enoic acid: [(2-propenoic acid, 3-iodo-, (2E)-)]
Organic Syntheses
(2003)
80
129
(doi: 10.15227/orgsyn.080.0129)
Development of β-keto 1,3-dithianes as versatile intermediates for organic synthesis
Organic & biomolecular chemistry
(2003)
1
15
(doi: 10.1039/b208982c)
A 2,3-butanedione protected chiral glycine equivalent—a new building block for the stereoselective synthesis of enantiopure N-protected α-amino acids
Chemical Communications
(2003)
3
468
(doi: 10.1039/b210673f)
- ‹ previous
- Page 53

